In this sweeping exploration, we’ll unpack the science, promise, setbacks, ethical debates, and realistic timelines behind one of medicine’s most tantalizing frontiers: bioprinted organs. From lab benches to hospital ORs, biomaterial innovation is rewriting what might be possible in replacing human donors.
Introduction: A World Strained by Donor Shortages
Across the globe, thousands die each year waiting for an organ transplant. In the United States alone, tens of thousands remain on waiting lists for kidneys, hearts, livers, and lungs that may never come. Statistics from transplant networks highlight that only a fraction of patients receive timely matches, and many perish before help arrives. Even when a donor organ is found, recipients often confront lifelong immune‑suppressing regimens to prevent rejection. These limitations have driven scientists, engineers, and physicians to ask a bold question: Can bio‑printed organs replace donors altogether?
Bioprinting represents a fusion of biology, engineering, and additive manufacturing — a technology so futuristic that it often sounds like science fiction. But underneath the hype lies serious research, real breakthroughs, and a global effort to make custom‑grown organs a reality.
What Is Bioprinting, Really?
At its core, bioprinting adapts 3D printing — building structures layer by layer — to living cells and biological materials (“bioink”). Unlike plastic or metal printing, bioprinting must maintain cellular viability, organize cells into functional architectures, and create scaffolds that mimic how organs naturally form in the body.
Different from traditional 3D printing, bioink often includes stem cells, growth factors, and biomaterials that support cell growth. The printer lays down these elements with precision that can reach micrometer scales — roughly the size of a single human cell. These structures can then mature in bioreactors or incubators, allowing cells to self‑organize and integrate.
Yet this elegant idea confronts enormous complexity: most organs are not simple blocks of tissue. They include vast vascular networks, multiple cell types, electrical conduction systems (in the heart), and intricate signaling pathways that have evolved over millions of years. Replicating even a fraction of this complexity poses deep scientific and engineering challenges.
How Close Are We Scientifically?
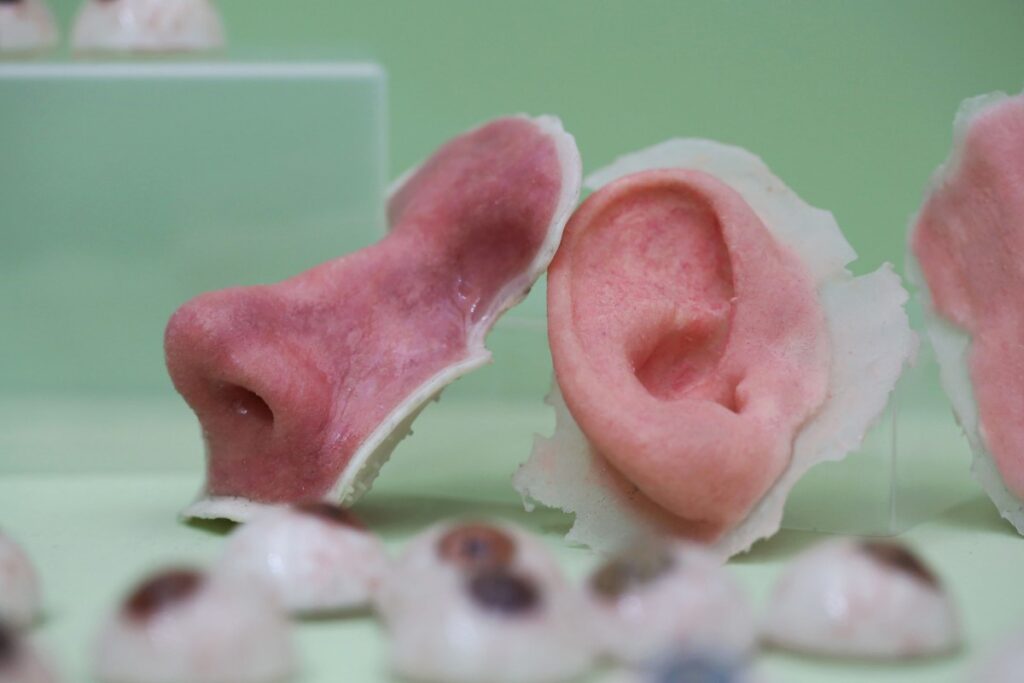
Early Wins: Tissue and Vascular Structures
Research labs worldwide have reported incremental but impressive achievements. Scientists have successfully bioprinted:
- Heart tissue patches that contract, although with weaker force than natural heart muscle.
- Blood vessel networks integrated into engineered cardiac tissue, bringing us closer to solving the critical blood‑supply problem.
- Miniature organoids that mimic the functions of larger organs at a simplified scale, enabling drug testing and disease modeling.
These breakthroughs illustrate not just proof of concept, but trajectories toward larger, more complex structures. They also reveal the iterative nature of progress — small biological modules build toward eventually assembling full organs.
However, while engineered tissues and organoids are functional for research, none are yet approved for large‑scale human transplantation. The challenge remains not just in printing tissue, but in ensuring that it truly integrates, survives, and functions long‑term inside a human body.
The Toughest Hurdle: Vascularization and Integration
One of the core roadblocks is vascularization: creating a network of blood vessels within a printed organ so every cell receives oxygen and nutrients. Without this, larger tissues die from a lack of blood flow. Researchers are exploring advanced printing techniques and sacrificial bioinks to generate channels that can later become vessels, but the process is far from solved.
Beyond vascularization, organs must integrate with:
- The recipient’s immune system.
- Innervation (nerve connections) for organs like the heart or lungs.
- Mechanical stresses (e.g., the pumping force of a heart or filtration pressure of a kidney).
These requirements push the technology beyond mere structural replication — toward fully functional physiology.
Timelines: When Might Bio‑Printed Organs Become Reality?
Predictions vary, partly because each organ presents unique obstacles. Analysts and researchers estimate broad windows:
- Simple tissues and organoids: already in use for drug testing and disease modeling.
- Proto‑organs and temporary grafts: possibly appearing in clinical research within the next decade.
- Fully functional, transplantable organs: often estimated to be 15–30 years away, depending on organ complexity.

Importantly, experts caution that timelines are estimates, not guarantees. Even with accelerated investment, regulatory approval processes, safety validation, and clinical trials are long and rigorous compared to incremental research milestones.
Beyond the Lab: Regulatory and Clinical Challenges
Turning bioprinted organs into a clinical reality requires navigating ethical, regulatory, and policy hurdles:
- Rigorous validation and safety testing: Demonstrating that printed organs are safe, effective, and durable inside human bodies.
- Standardization of manufacturing: Ensuring reproducibility, quality control, and scalable production.
- Clinical trial frameworks: Designing trials that ethically and effectively assess outcomes in human patients.
Regulatory agencies like the U.S. FDA, EMA, and counterparts worldwide have yet to establish comprehensive frameworks specifically for living, printed organs — a gap that adds complexity to timeline projections.
Ethical, Societal, and Economic Imperatives
Bioprinting isn’t just a technical challenge — it’s a societal one. Ethical questions swirl around access, equity, and impact:
- Who gets priority? If printed organs are expensive, will wealthy patients receive them first?
- Health disparities: Could this technology deepen global inequalities in healthcare?
- Philosophical concerns: What does it mean to grow life, engineer bodies, and redefine organ donation?
Traditionally, organ donation relies on the goodwill of donors and their families — a deeply human, altruistic system. Bioprinted organs, by contrast, could be commodified, automated, and subject to market forces.
While these ethical questions don’t block innovation, they must inform policies and investment decisions. Without a proactive framework, we risk amplifying inequality and misunderstanding rather than empowering patients.
The Ripple Effects Beyond Transplants
Even if fully bioprinted organs are years away, the surrounding technologies are already transforming medicine:
- Pharmaceutical development: Organoid tissues offer better models for drug efficacy and toxicity.
- Disease modeling: Bioprinted tissues help researchers study conditions like cancer, fibrosis, and genetic disorders.
- Reducing animal testing: The UK and other nations are exploring printed human tissues to replace animal models in safety testing.
The broader “biofabrication” revolution encompasses tools, platforms, and models that benefit medicine long before full organ replacement becomes routine.
Looking Ahead: A Balance of Hope and Realism
So, can bio‑printed organs replace donors soon? The honest answer is both yes and not yet:
Yes — because the science is real, progressing, and opening unprecedented doors in regenerative medicine. Already, bioprinted tissues are proving their worth in research, testing, and early clinical applications.
Not yet — because the complexity of human organs, regulatory pathways, and real‑world integration means that fully functional, transplantable organs replacing human donors remain a long‑term prospect.
The next 30 years may see a radical shift in how we approach organ failure, one that moves from waiting for donors to engineering organs on demand. But that future will unfold through careful science, thoughtful policy, and ethical foresight — not merely technological hype.
