Introduction
Imagine a device so capable and intimate that it knows you better than you know yourself—tracking your body’s chemistry in real time, predicting disease before symptoms surface, and guiding personalized health decisions minute by minute. This isn’t science fiction. With the rapid evolution of implantable sensors that measure biomarkers inside the body, this vision is accelerating toward reality. Yet it also opens profound questions about autonomy, privacy, safety, and what it means to be “connected” to technology at the most intimate level.
In this article, we’ll explore the science, the potential, the challenges, and the ethical dilemmas of implanting a sensor that could measure every biomarker in your body. Along the way, we’ll unpack how such sensors work, what they could monitor, why they matter, and what we need to consider before adopting them.
What Is a Biomarker—and What Could “Every Biomarker” Mean?
A biomarker is any measurable biological indicator of a physiological or pathological process. Biomarkers can range from simple metrics like glucose and pH, to more complex proteins related to inflammation, immune response, or cancer progression. They are used in diagnostics, monitoring disease progression, evaluating treatment effectiveness, and even predicting future health events.
An implantable sensor that measures “every biomarker” wouldn’t literally measure every possible biomarker known to science—that list could be thousands long—but it could target many key molecules and physiological variables simultaneously, offering a multidimensional, continuous picture of health.
How Implantable Biomarker Sensors Work
At the heart of biomarker monitoring are biosensors—devices that detect biological molecules and convert that detection into measurable signals. These can be electrochemical, optical, mechanical, or based on field-effect transistors like ISFETs (ion-sensitive field-effect transistors).
Implantable sensors are designed to reside within the body and measure biomarkers directly from blood, interstitial fluid, or tissues. They typically consist of:
- Recognition elements – molecules such as enzymes, antibodies, or aptamers that bind specific biomarkers.
- Transducers – components that convert the binding event into an electrical signal.
- Communication modules – wireless systems that transmit the data to external devices.
Modern research focuses on making these sensors smaller, biocompatible, power-efficient, and capable of continuous long-term monitoring without frequent calibration or replacement.
Example: Continuous Protein Monitoring
Traditionally, continuous monitoring was limited to small molecules like glucose. However, recent breakthroughs show sensors that can dynamically monitor protein biomarkers linked with inflammation—a key indicator in many chronic diseases like heart disease and cancer.
This kind of monitoring has massive implications. Inflammation isn’t just a local symptom—it’s a systemic signal of immune activity and disease progression.
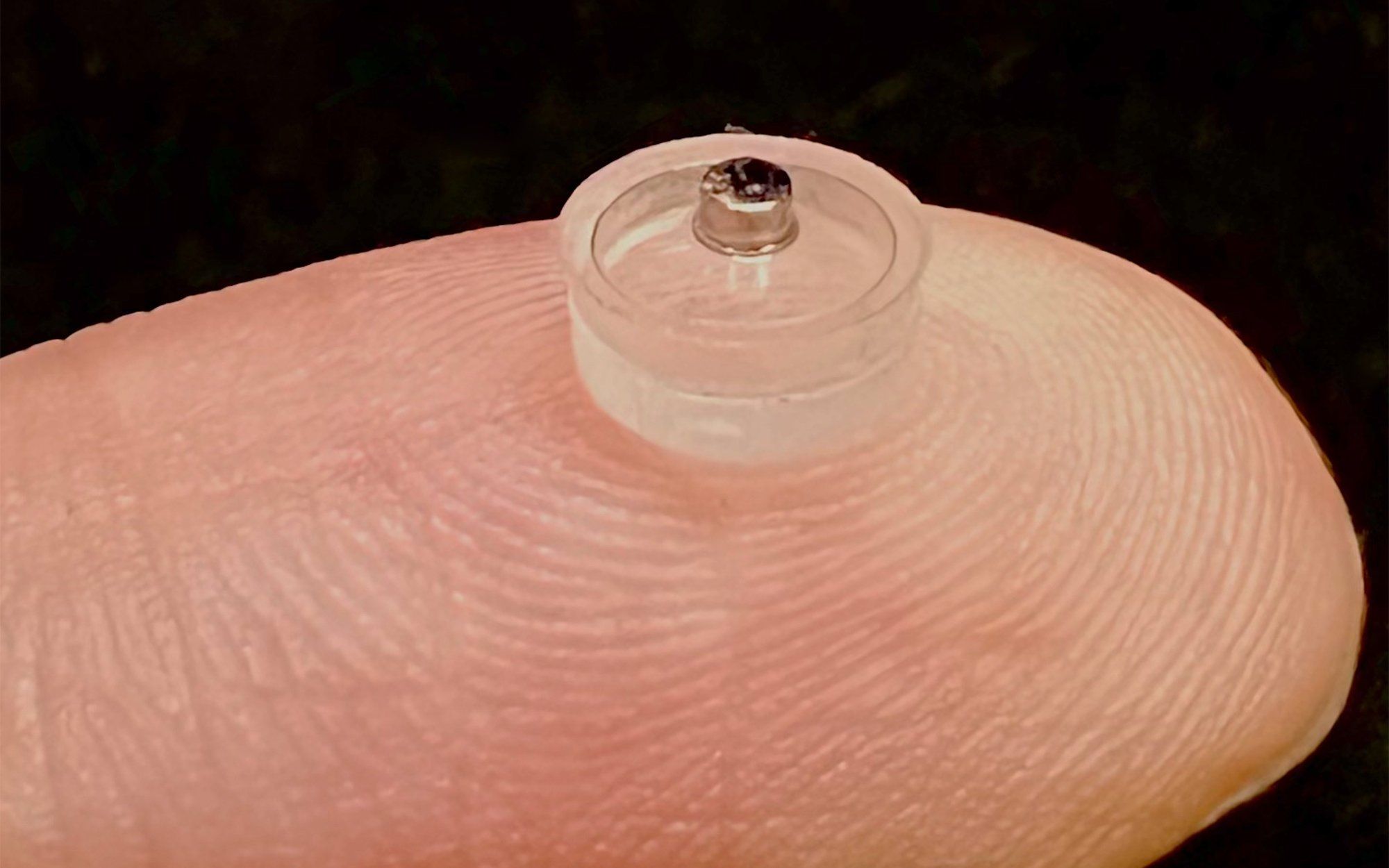
Example: Implantable Glucose Sensors
One of the earliest and most developed implantable sensors is the continuous glucose monitor. Long-term internal glucose sensors have been demonstrated, capable of providing real-time glucose data for extended periods.
This proves the principle: an internal detector can reliably operate inside the body, collect data, and transmit it wirelessly.
The Potential Benefits
Personalized, Predictive Health
Instead of episodic snapshots from annual checkups or occasional lab work, implantable sensors could provide an unbroken timeline of biomarker dynamics. Fluctuations in hormone levels, inflammation markers, metabolic substrates, or immune responses could signal early disease or reveal patterns tied to lifestyle, sleep, stress, or nutrition.
This kind of granular data could power:
- Early detection of disease
- Tailored medical interventions
- Customized nutrition and fitness plans
- Dynamic medication dosing
- Better understanding of aging processes
Real-Time Monitoring of Chronic Conditions
Chronic diseases like diabetes, cardiovascular disease, autoimmune disorders, and kidney disease could be monitored in real time. This would allow clinicians, and potentially patients themselves, to adjust therapy proactively rather than reactively.
Less Invasive Diagnostics
Rather than demanding frequent blood draws or biopsies, a sensor internally captures and reports data without repeated invasive procedures.
Integration with AI and Predictive Analytics
Imagine pairing continuous biomarker streams with powerful AI models that can identify patterns or early signs of disease before overt symptoms occur. This is the core promise of precision medicine—medicine tuned to the individual’s biology, not broad population averages.
The Technical Challenges
Despite progress, translating sensor research from lab to clinic remains hard.
Biocompatibility and Immune Response
The body tends to reject foreign objects. Sensors need coatings or materials that minimize inflammation and fibrotic encapsulation. This is an ongoing area of research and innovation.
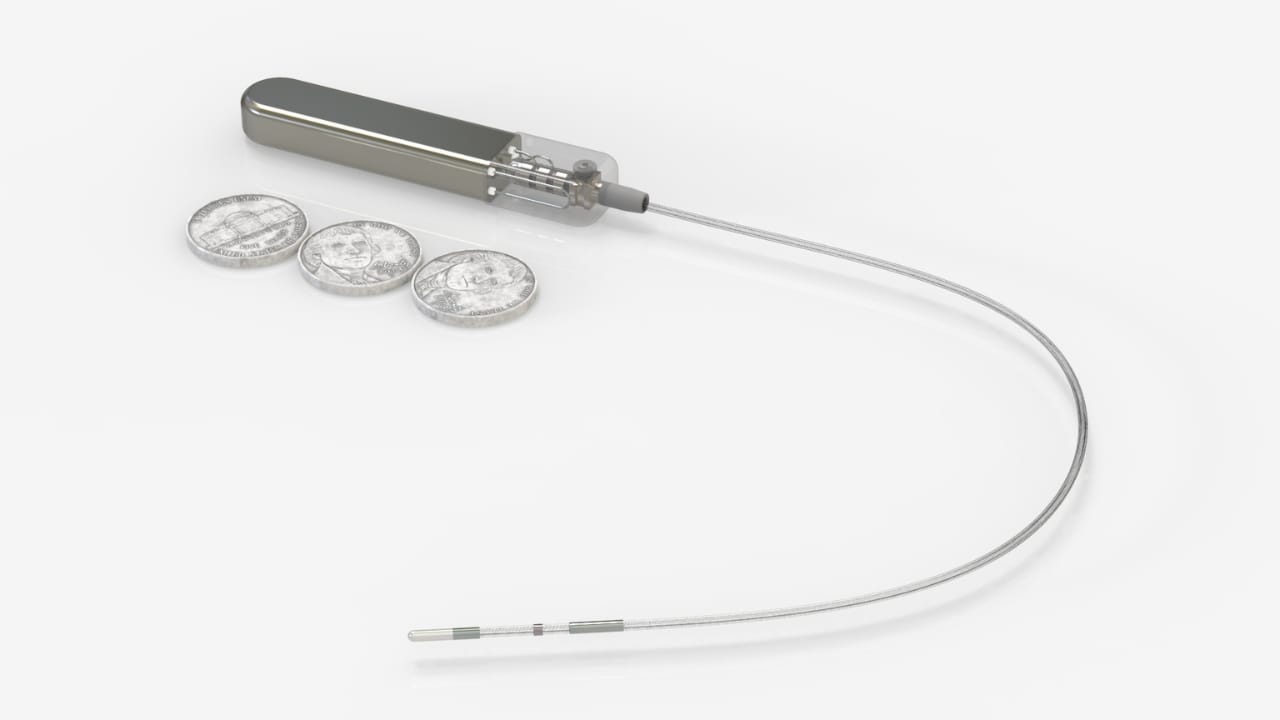
Power and Data Transmission
Every implanted device needs power. While some use batteries, others explore wireless or inductive power transfer. Data transmission must be secure, robust, and require minimal power—especially in a body where energy harvesting is nontrivial.
Long-Term Stability and Reliability
Sensors must operate reliably for years, not just weeks. This means resisting biofouling, maintaining sensitivity, and minimizing drift. These remain significant engineering challenges.
Calibration and Accuracy
Implants must maintain accuracy over time, despite complex and fluctuating biochemical environments. Continuous recalibration using internal or external references could help—but adds complexity.
The Ethical and Social Considerations
Even if technically perfected, the true question may be social: Should we implant devices that “know us” at the molecular level?
Data Privacy and Security
Continuous biomarker data will be deeply personal. Who owns that data? Who has access? Could insurers or employers misuse it? Establishing rigorous data governance and encryption standards is paramount.
Autonomy and Consent
Patients must fully understand what they’re agreeing to—especially for long-term implants with permanent physiological access.
Inequality and Access
Who will benefit? Will such technology widen health disparities if only wealthy individuals or privileged healthcare systems can afford it?
Psychological Impact
Knowing every fluctuation in your body could be empowering—or anxiety-inducing. How much information is too much?
The Future Landscape
The field of implantable biosensors is progressing rapidly, backed by research, start-ups, and major medical institutions. Sensors capable of tracking glucose, pH, mechanical strain, and protein levels are already being tested and deployed in controlled settings.
As multi-modal sensing platforms emerge—integrating electrical, optical, and biochemical detection with AI-driven models—the possibility of a holistic internal health dashboard becomes clearer. This fusion could not only change medicine but transform how we think about health and life itself.
We’re advancing toward a future where biological data flows continuously, where health is measured in streams not snapshots, and where the line between organ and instrument blurs. But whether society embraces body-implanted biomarker systems will depend not just on science, but on values, ethics, and collective choices.
